 The IRB reviews and approves all research involving human participants prior to the research being conducted. This includes research conducted by faculty, staff and students, regardless of location or funding.
The IRB reviews and approves all research involving human participants prior to the research being conducted. This includes research conducted by faculty, staff and students, regardless of location or funding.
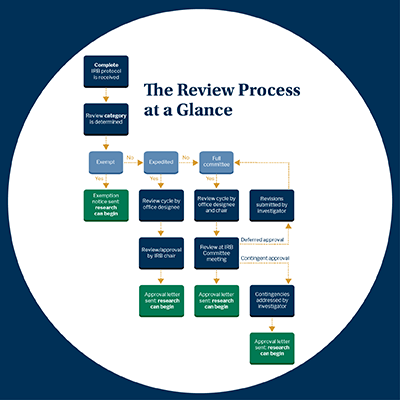
Review Timeline: Applications are typically reviewed within 30 days of submission. However, expedited or full reviews with minimal or greater risk are more complicated, may require multiple reviewers, and could result in an extended review period.
Requests for revisions or additional information will be sent via email by the IRB Chair. Students should work with their faculty advisor(s) to respond to the IRB's requests.
Research (including data collection) may not begin until the proposal is successfully reviewed and approved. Upon determination, an IRB approval letter with appropriate review designation is sent to the applicant’s email. If you have any questions, please consult with your faculty advisor and, if appropriate, email the IRB at irb@endicott.edu.
Note: Exempt research does not mean "No review is necessary."
More information is available in Canvas (login required):
- IrbNet instructions (the new online proposal system effective 9.9.2020)
- New student IRB application form
- New Faculty Sponsor and Student PI IRB Manual
- Preliminary Zoom guidelines (August 19, 2020)
Helpful Links
Do You Need IRB Review?
What is the Difference Between Anonymity and Confidentiality?
Faculty Research Using Students as Participants
Key Points for New Common Rule
Review Types
Consent Statements & Debriefing Statement Samples
Survey Tools
Ethical Resources & CITI Training

